 
We have not dissociated liquid water into ions rather, we wrote H 2O (l). If any of the products is in liquid, solid, or gaseous form, we leave the compound as it is.įor example, as we did in the last two sample problems. We can only dissociate the aqueous compounds. Determine the spectator ions.įollowing the same instructions as in the previous problem we get:ĬaCl 2 (aq) + Na 2CO 3 (aq) → CaCO 3 (s) + 2NaCl (aq)Ĭa + (aq) + 2Cl - (aq) + 2Na + (aq) + CO 3 2- (aq) → CaCO 3 (s) + 2Na + (aq) + 2Cl - (aq) Write molecular, complete ionic, and net ionic equations for the reaction between calcium chloride and sodium carbonate. Na + and Cl - ← spectator ions Sample Problem Step 4: determine the spectator ions by stating the ones that have been omitted from the net ionic equation H + (aq) + OH - (aq) → H 2O (l) ← net ionic equation Step 3: Exclude the ions that remain unaltered on both sides of the equation H + (aq) + Cl - (aq) + Na + (aq) + OH - (aq) → Na + (aq) + Cl - (aq) + H 2O (l) ← complete ionic equation HCl (aq) + NaOH (aq) → NaCl (aq) + H 2O (l) → balanced molecular equation Step 1: Determine the molecular formulas for the given substances and expected products to write the molecular equation. Write a molecular, complete ionic, and net ionic equation for the reaction between hydrochloric acid and sodium hydroxide. 
Spectator ions are ions that have been excluded from the net ionic equation.Īs we discussed the major definitions, let’s consider several specific examples of ionic equations. The net ionic equation is a shortened version of the complete ionic equation since we omit the ions that are unaltered on both sides of the equation. The complete ionic equation is a balanced chemical equation that involves component ions of the ionic compounds. The molecular equation is a balanced chemical equation that involves ionic compounds but in the form of molecules. We should define several terms before we continue with specific ionic equations. Read more about Compounds, Formulae, and Equations Ionic EquationsĪs we already discussed in the article, we can write an ionic equation for every reaction which involves aqueous reactants. Insoluble bases DO NOT dissociate Dissociation of salts 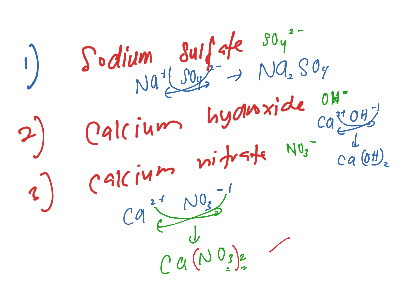
Soluble bases (alkali bases) dissociate releasing metal cations and hydroxyl anions Overall dissociation: H 2SO 4 (aq) → 2H + (aq) + SO 4 2- (aq) Dissociation of bases Step II: HSO 4 - (aq) → H + (aq) + SO 4 2- (aq) Step I: H 2SO 4 (aq) → H + (aq) + HSO 4 - (aq) Let’s consider dissociation of different substances: Dissociation of acidsĪcids consisting of 1 hydrogen atom dissociate completely in one step:Īcids consisting of 2 or more hydrogens dissociate in steps In reality, we have H 3O + in a solution as a result of ionic dissociation of hydrochloric acid, but for simplicity, we just write H + instead of H 3O +. Let’s consider the simple dissociation of an acid: Since water is a polar molecule, it is considered a dipole.Įven though we denote ions as X + or Y -, we have hydrated ions in the solution, meaning that water molecules surround the ions. Typical examples are – DI water, ethanol, sugar (C 6H 12O 6 – glucose), some other organic compounds, etc.Įlectrolytic dissociation, also referred to as ionic dissociation, occurs when an electrolyte is dissolved in an aqueous solution (meaning that it is dissociated into ions).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
